Project B1 | Dendritic polyelectrolytes to promote enhanced neuronal biocompatibility
Block / Haag / Kennedy
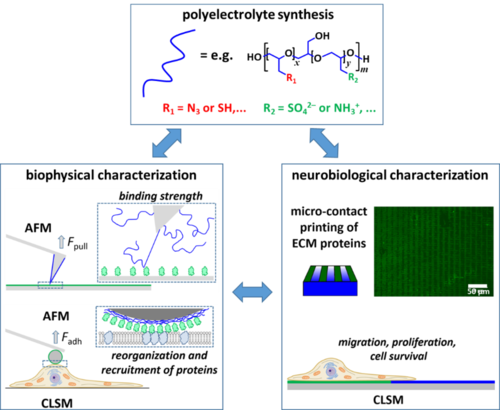
The development of advanced prosthetics for the nervous system is evolving rapidly. However, little attention has been paid to extending neural biocompatibility beyond conventional metal and silicon electrodes. The development of engineered extracellular matrix (ECM) that is compatible with the nervous system has the potential to revolutionize the capacity to implant technology into the nervous system. Synthetic multivalent hyperbranched polymers modified with anionic functional groups (sulfate groups, negative at neutral pH) or amine (positive at neutral pH) terminal moieties can be used to coat surfaces or prepare 3D gel formulations. By combining approaches from macromolecular chemistry, biophysics, and neurobiology, project B1 aims to optimize these coatings and gels to function as synthetic extracellular matrices with enhanced biocompatibility in the central nervous system.
To achieve this goal, the design of the synthetic ECMs will employ a varying architecture and functionalization of the synthesized polymers. Of particular importance are ECMs that have been functionalized with proteins known to promote neural cell survival and/or proteins known to be important for the attachment and migration of neurons, such as netrin-1. The interaction of synthesized polymers with functional CNS proteins will be investigated on the molecular level using atomic force microscopy (AFM)-based dynamic force spectroscopy and flow-based force measurements. This will provide information on the energy landscape of the protein-polymer interaction and the mechanical binding strength. These measurements will be further complemented by equilibrium binding studies such as ITC and SPR to obtain the underlying binding energetics. On a cellular level, it will be investigated how synthetic ECMs based on multivalent heparin mimetic polyanions bind and locally concentrate trophic factors and netrin-1. These measurements will be complemented by colloidal probe AFM experiments, in which a µm-sized bead is coated with protein-functionalized synthetic ECMs and brought into contact with the cell surface. Combining atomic force and fluorescence microscopy will enable monitoring the local reorganization and recruitment of proteins at the cell membrane and allow correlation of such recruitment processes with changes in cellular mechano-adhesion to the ECM-coated bead. These experiments will allow us to probe the ECM-cell interactions in a bottom up approach, gradually increasing the complexity of the investigated systems, and thus enabling the design of synthetic ECMs with enhanced neural biocompatibility.
Subjects: Biochemistry | Biophysics | Neuroscience